0.2μm AseptiCap® WL/WS-γ 25mm, 50mm
mdi AseptiCap® WL/WS-γ are low protein binding hydrophilic PVDF gamma sterilizable membrane inline capsule filters, designed for sterile filtration of very small fluid volumes in formulation and process development labs.
These capsule filters are validated to meet compendia and regulatory requirements and are well characterized. They meet key process requirements such as absolute retention efficiency, extremely low extractables, high throughputs, wide chemical compatibility and other important characteristics.
- Absolute retention
- 100% integrity tested
- Low protein binding
- Low extractables
- Very low hold up volume
Sterile Filtration of:
- Antibodies
- Protein Solutions
- Buffers
- Heat labile additives
- Vaccine concentrates
- Small Volume Parenterals
| Construction | |||
| Membrane | 0.2µm Hydrophilic PVDF | ||
| Prefilter Membrane | 0.8 µm or 0.45 µm Hydrophilic PVDF | ||
| Plastic parts | Gamma Stable Polypropylene | ||
| Size | |||
| Size |
|
||
| Effective Filtration Area (Nominal) |
|
||
| Dimension (End to End) - Female Luer Lock Inlet/ Male Luer Slip |
|
||
| Dimension (End to End) - ¼" SHB |
|
||
| Dimension (End to End) - ¾" Sanitary Flange Inlet I/O |
|
||
| Operational Radius (with Vent/ Drain) |
|
||
| Integrity Testing / Retention | |||
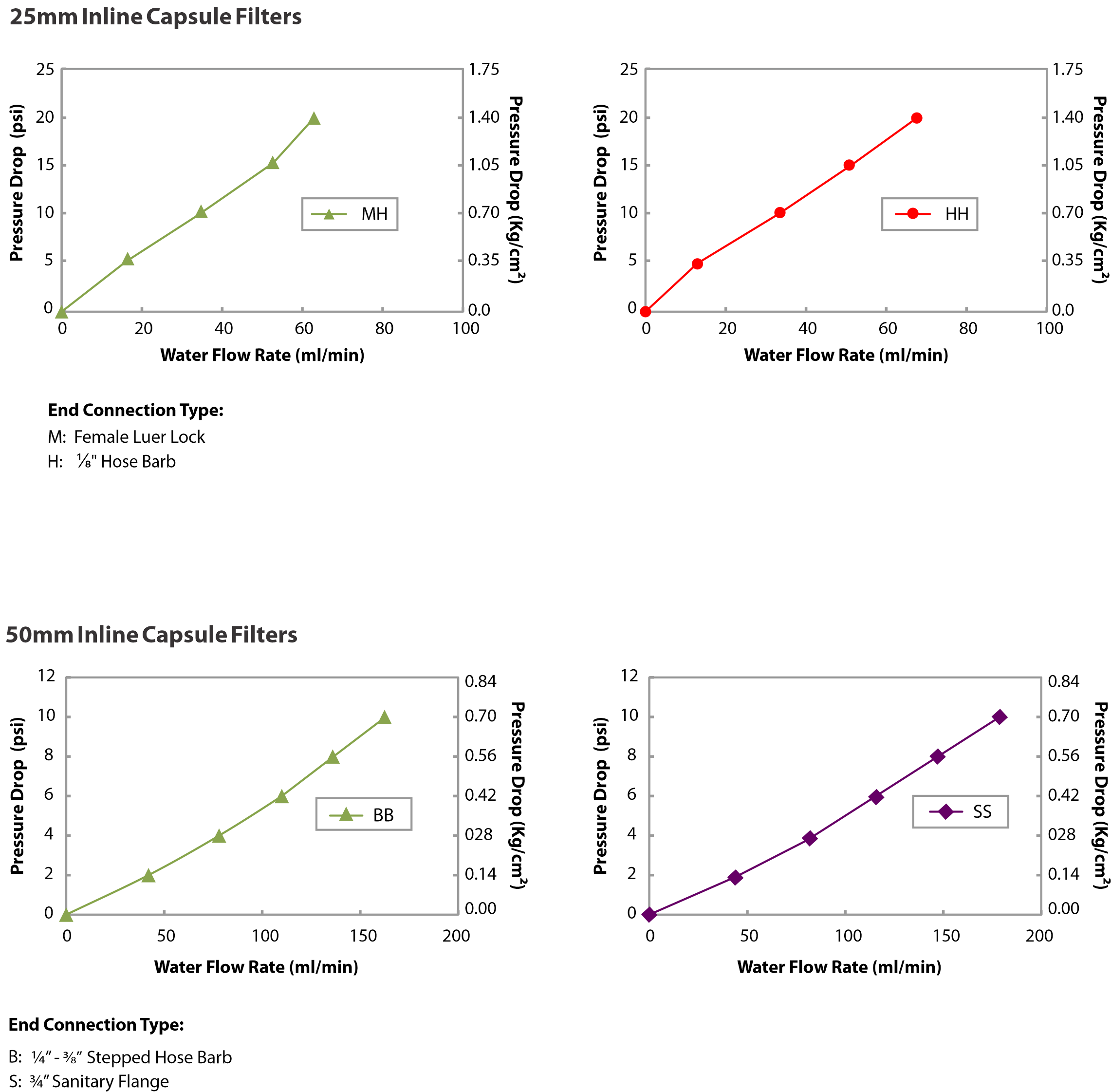
| Bubble Point | > 50 psi (3.51 Kg/cm² ) with Water | ||
| Bacterial Retention | LRV >7 for Brevundimonas diminuta (ATCC 19146) per cm² | ||
| Operational | |||
| Max. Operating Temperature |
|
||
| Max. Differential Pressure |
|
||
| Sterilization By Irradiation | Gamma Irradiatiable up to 50 kGy. These filters must not be autoclaved or in-line steam sterilized. | ||
| Shelf Life | 2 years after gamma sterilization | ||
| Assurance | |||
| Toxicity | Passes Bioreactivity test, In Vivo, as per USP <88> for Class VI plastics | ||
| Cytotoxicity | Passes Biological Reactivity Tests, In Vitro, USP <87> for cytotoxicity | ||
| Bacterial Endotoxin | Aqueous extracts exhibit < 0.25 EU/ml as established by Limulus Amebocyte Lysate (LAL) Test as per USP <85> | ||
| Bioburden | Bioburden level is < 1000 cfu/filter device as per ISO 11737-1 : 2018 | ||
| Non Fiber Releasing | Passes test as per USP and comply with USFDA 21 CFR Part 210.3(b)(6) for fiber release | ||
| TOC and Conductivity | Meets the WFI requirements of USP for TOC <643> and Conductivity <645> after a 500ml flush for inline filters | ||
| Particle Release | The filtrate complies with USP <788> test for particulate matter in injections. | ||
| Indirect Food Additive | All Polypropylene components meet the FDA Indirect Food Additive requirements cited in 21 CFR Part 177.1520 | ||
| Extractables with WFI | Passes NVR test as per USP <661> | ||
| Oxidizable Substances | Passes test as per USP <1231> | ||
| Quality Management System | ISO-9001 Certified | ||
| USFDA | DMF No. 015554 | ||
AseptiCap® WL/WS- γ (25mm)
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||
* Gamma sterilized filters cannot be gamma irradiated again
Example:
| IWSX | 06 | 01 | M | N | R | X | 1 | 04 |
AseptiCap® WL/WS- γ (50mm)
|
|
|
|
|
|
|
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
*¼" Stepped Hose Barb and ¾" Sanitary Flange are available in filters with vent only
**Female luer lock is available in filters without vent only
Example:
| VWSX | 10 | 01 | SS | SS | R | X | 1 | 04 |
| Datasheet | Download |