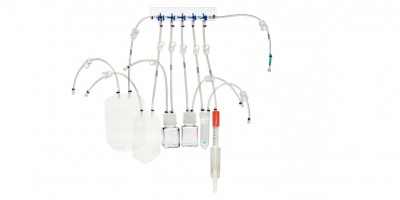
ASESS® Aseptic Sampling Systems
mdi ASESS® are gamma irradiated single use disposable systems for aseptic fluid sampling at various stages in pharmaceutical and biopharmaceutical processing. These systems minimize chances of false positives and reduce cleaning requirements over traditional sampling systems.
Aseptic sampling of:
- Sterile bulk and sterile transfers
- Vaccine formulations at different maturation steps
- Fermentation cycles
- Cell culture from bioreactors
| Materials of Construction | |||
| Septum | Platinum Cured Silicone | ||
| Needle | 316L SS | ||
| Bag Film | Multilayered Film type FBG-1 | ||
| Tubing |
|
||
| Operational | |||
| Operating Pressure | 5 psi | ||
| Steam in Place (Sanitary flange adaptor assembly) | 125ºC for 30 min | ||
| Operating Temperature Range | -20ºC to 60ºC | ||
| Assurance | |||
| Integrity | 100% Integrity tested | ||
| Non Animal Origin | Comply with the requirements laid down in the “Note for Guidance EMEA/410/01, rev. 03”for inputs of Non-Animal Origin | ||
| Sterility | Gamma Irradiated | ||
Sampling Systems
Gamma Sterile ASESS® Aseptic Sampling Systems with 2mm Needle
| A044XXXXXXSS301 | 5x100 mL sampling bags and 1x250 mL drain bag |
| A023XXXXXXSS301 | 10x100 mL sampling bags and 2 x 250 mL drain bags |
| A028XXXXXXSS301 | 1x100 mL sampling bag |
| A029XXXXXXSS301 | 1x250 mL sampling bag |
Gamma Sterile ASESS® Aseptic Sampling Systems with 1mm Needle
| A1359XXXXXSS301 | 5x250 mL sampling bags and 1x250 mL drain bag |
| A1360XXXXXSS301 | 1x100mL sampling bag |
Accessories
Sampling Port
| ASPC25XXXXXXX01 | 25mm sampling port connection for both 1mm or 2mm needle, single sampling port |
| ASPCIG02XXXXX01 | Ingold Adapter - Sampling port connection for both 1mm or 2mm needle, two sampling ports |
| ASPCIG02XXLXX01 | Ingold Adapter for Longer Ingold Ports- Sampling port connection for both 1mm or 2mm needle, two sampling ports |
| ASPC5005XXXXX01 | 50mm sampling port connection for both 1mm or 2mm needle, five sampling ports |
Crimping Tool
| CTLXXXXXXXXXX01 |
CrimGard Crimped Tube Cover
| CGCXXXXXXXXXX02 for pack size of 10 |
ASESS® Port Blind
| AASSBXXXXXXXX02 for pack size of 10 |
Customization
ASESS® Aseptic Sampling Systems can be customized to suit user requirements regarding tubing sizes, sizes, sampling ports, and vessel port. The technical feasibility of the required design is established based on available components and an initial drawing is proposed. Products prototyping and final approval lead to customized system realization.
Validated for:
- Sterility as per USP <71>
- Class VI Plastics as per USP <88>
- Cytotoxicity as per USP <87>
- Bacterial Endotoxin <0.25EU/ml
- TOC as per USP <643>
- Conductivity as per USP <645>