We are witnessing an industry-wide shift in biopharmaceutical manufacturing processes from reusable stainless steel systems to single use disposable systems, due to the need for higher flexibility, faster turnaround time and lower documentation and energy costs.
Since Single Use Systems (SUS) are customized multi-component polymeric assemblies, this shift has resulted in new challenges for the bioprocess owners with regards to leachables, biosafety, sterility, integrity and particulate matter.
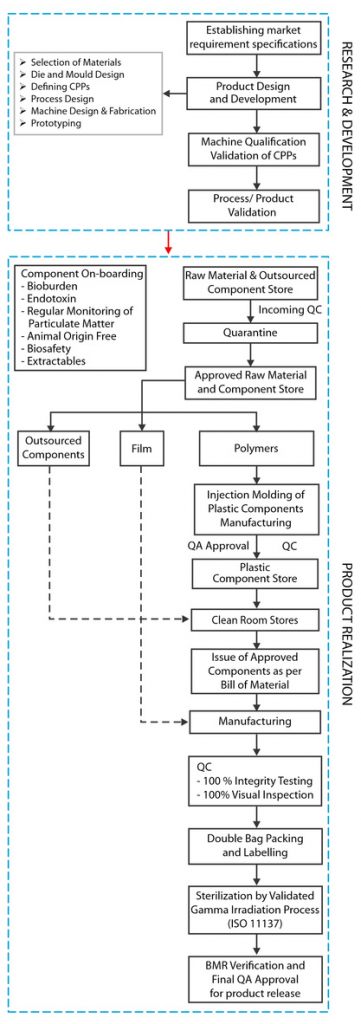
The bioprocess owner as an end user, usually does not have the infrastructure or expertise to verify these single use assemblies for such regulatory and functional concerns and to ensure compliance. It has thus become imperative for the SUS manufacturer/supplier to address these challenges through a well developed system that assures quality at every step of design, development and manufacture of these single use fluid management systems.
At mdi we corroborate this by incorporating the elements of Quality by Design at every step, starting with defining the Quality Target Product Profile (QTPP) by establishing market requirement specification (MRS).


