The growing trend towards incorporation of single use technologies into bioprocesses as an alternative to reusable systems have paved the way for many single use components and systems as these provide increased manufacturing flexibility.
Bioprocess operations such as cell-culture and mixing involve disposable systems which require real time monitoring to determine whether various critical process parameters (CPPs) such as pH, conductivity, dissolved oxygen, etc are maintained within specifications.
To measure CPPs, different types of on-line sensors are used. On-line sensors interface directly with Bioreactor/Mixer and are in contact with contents in it. Such systems are also available with pre installed single use sensors. However, these disposable sensors have multiple challenges in terms of accuracy, reliability, on site calibration, etc.
These disposable sensors also reduce the overall residual shelf life of high value Single Use Mixers/ Bioreactors, making reusable sensors the preferred choice. The reusable sensors can be calibrated and autoclaved before use.
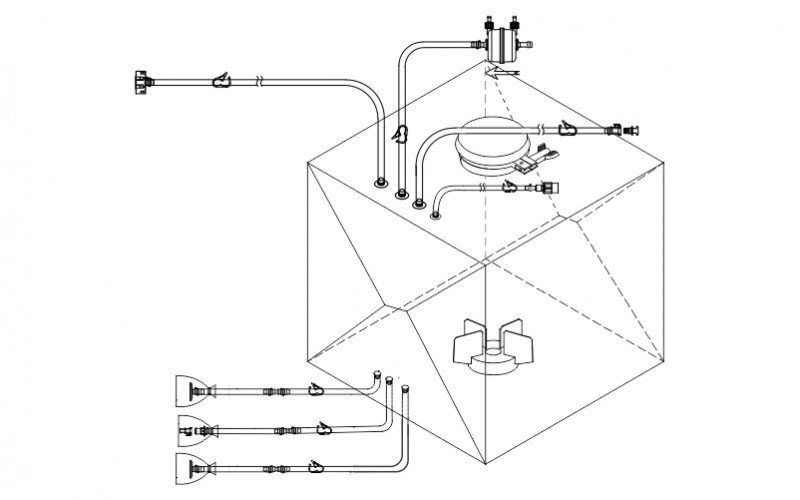
MDI AseptiProbe Bellow Assembly is designed to address the need to aseptically connect reusable sensors to single use bioreactors or mixers. These assemblies enable real-time monitoring while eliminating laborious cleaning validation requirements, reducing the risk of cross-contamination, and improving sterility assurance. These Bellow Assemblies are validated to prevent microbial ingress and are 100 % integrity tested.
Aseptiprobe Bellow assemblies are manufactured in GMP facilities with ISO clean room areas and comply with various international standards and guidelines such as USP, ISO and CFR for biosafety, bioburden, bacterial endotoxins, and particulate matter.