Bio-pharmaceutical manufacturing is a complex, multistep process involving a very wide variety of process streams under different process conditions at different steps. These process streams include cell culture media, media additives, growth regulators in the upstream and post centrifuge cell harvest supernatants, post viral inactivation process intermediates, buffers, and high value product concentrates in the downstream. Filtration and purification of such a wide spectrum of process streams, to achieve different process objectives at each process step, is quite a challenge for the process owner. Also the increasingly competitive market conditions, due to the introduction of biosimilars, further escalates the demand for higher standards of filter performance, with respect to economy, safety, and impact on final product quality.
Sterilizing Filters in Bio-pharma
Filtration and purification in bio-pharmaceutical processes involves high value chromatography, ultrafiltration, micro-filtration equipment as well consumables. Micro-filtration accounts for a very high (approximately) 25% of the total cost. The 0.2μm sterilizing grade filters are a huge component of the total micro-filtration needs and are critical for the success of multiple process steps in bio-pharmaceuticals. These play a central role not only in sterile filtration of final product concentrates but are widely used in nearly every step of bio-pharmaceutical production processes. They are used as protective filters to highly expensive virus filters, and chromatography columns against premature blocking by cell debris, precipitate fines etc, at various stages.
In addition, sterile filters perform the important task of controlling microbial load throughout the process chain and of protecting bulk products from contamination by microorganisms during intermediate storage.
Fig.1 Highlights the need for sterilizing grade filters at different process steps in a typical bio-pharmaceutical production of monoclonal antibodies.
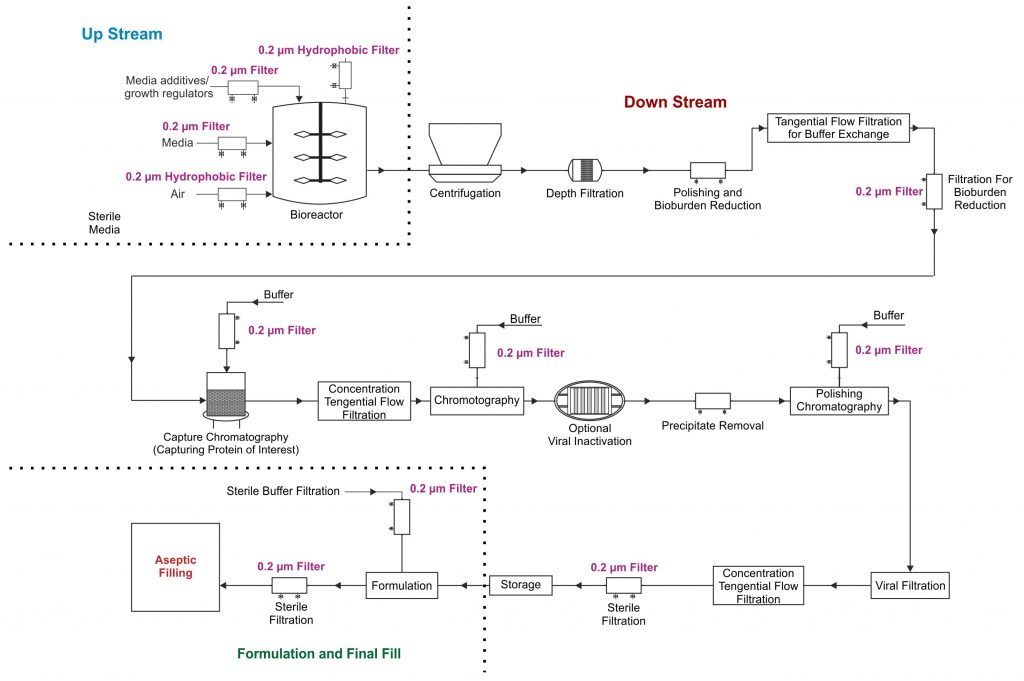
0.2μm sterilizing grade filters are high value validated micro-filtration devices and process owners use a variety of pre-filters to protect these and to make the overall filtration process more efficient. Some of the process streams are extremely difficult to filter and require use of highly efficient pre-filters. However fouling agents such as colloids, lipids, precipitates and cell debris fines, can penetrate even the most efficient pre-filters in small quantities but large enough to plug the 0.2μm sterilizing filters.
A typical case is mammalian cell expression system for monoclonal antibodies resulting in higher cell density and the consequent lower cell viability with increased cell fragments and cell organelles. These tend to pass through conventional primary and secondary clarification systems and may result in relative faster choking of sterilizing filters. This has resulted in a much larger footprint for the sterilizing filters and therefore much increased costs.
It thus becomes nearly impossible to warrant that a batch of a specific stream at a specific process step could be processed through the existing sterilizing grade filters and a need to accommodate change out of filters mid-batch and in some cases having to isolate a complete batch because it could not be processed can arise more often than acceptable. The process owners are thus always looking for high throughput (total filtered volume) sterilizing filters.
AseptiPrime KS: A revolutionary new asymmetric PES membrane filter
AseptiPrime KS with outstanding fluid handling capacities and consequently much higher throughputs can help overcome these challenges and result in a dramatic improvement in process economies due to much lower aseptic filtration footprint.
Comparative Throughput Analysis
Throughput studies were conducted with commercially available PES membrane capsule filters from leading micro-filtration brands and compared with MDI AseptiPrime KS capsule filters. All test filters were double layered serial filtration devices. The fluid streams selected were representative of the wide variety of buffer solutions and media used in bioprocesses based on mammalian cell as well as microbial expression systems.
Table 1. Shows difficult to filter buffer solutions used in bioprocesses
| Buffers | Lot No. |
|---|---|
| 1M Arginine | 0000198135 |
| 6M Urea | 457468091 |
| Guanidine HCL | 1571B90 |
Table 2. Shows different culture media to study microbial and mammalian cell expression
| Expression System | Culture Media | Lot No. |
|---|---|---|
| Mammalian Cell Culture | Soy media | SOY0011812 |
| RPMI media | RPM0012112 | |
| Microbial Cell Culture | Luria Bertani | 0000230874 |
| Yeast Extract Peptone Dextrose(YPD) | 0000203890 |
The representative process streams were filtered at a constant pressure and volume till 80% flow rate decay was recorded. Based on the test results, membrane permeability for each filter tested was calculated and estimations of the required number of equivalent 10 inch filter cartridges were made for a 10,000 L batch of buffer filtered in 1 hour.
Fig.2 & 3 Represent the comparative performance of different sterilizing filters for culture media used for mammalian cell expression.
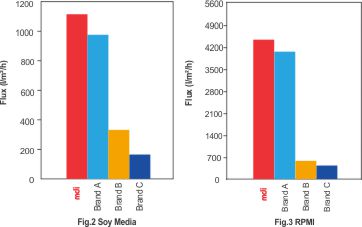
Fig.4 & 5 Represent the comparative performance of different sterilizing filters for culture media for microbial cell expression.
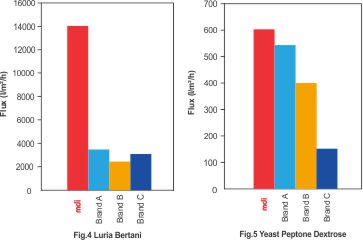
Fig.6, 7 & 8 Represent the comparative performance of different sterilizing filters for different buffer solutions.

Table 3. Shows number of 10” Capsule filters required for filtration of 10,000 L batch in 1 hour.
| Solutions | MDI | Brand A | Brand B | Brand C |
|---|---|---|---|---|
| Soy Media | 15 | 17 | 50 | 91 |
| RPMI | 9 | 7 | 35 | 48 |
| Luria Bertani | 1 | 5 | 6 | 5 |
| YPD | 28 | 31 | 42 | 107 |
| 1M Arginine | 12 | 16 | 29 | 51 |
| 6M Urea | 33 | 50 | 98 | 123 |
| 6M Guanidine HCl | 12 | 24 | 20 | 23 |
Conclusion
MDI AseptiPrime KS PES membrane filters consistently outperform other available filters from leading microfiltration brands. MDI AseptiPrime KS exhibit much higher throughput (approximately 40-100% more) when compared to other brands and resulting in much smaller footprints and much enhanced economics.

