Closed loop sterility testing systems have been successful in prevention of false positives while sterility testing of sterile injectables. However, there are special containers such as insulin cartridges (that fit into hand held delivery systems), wherein increased manual intervention is required for pooling the same into a 100ml vial before these can be transferred/filtered through the closed sterility test canisters. This increases the possibility of extraneous contamination and thereby false positives.
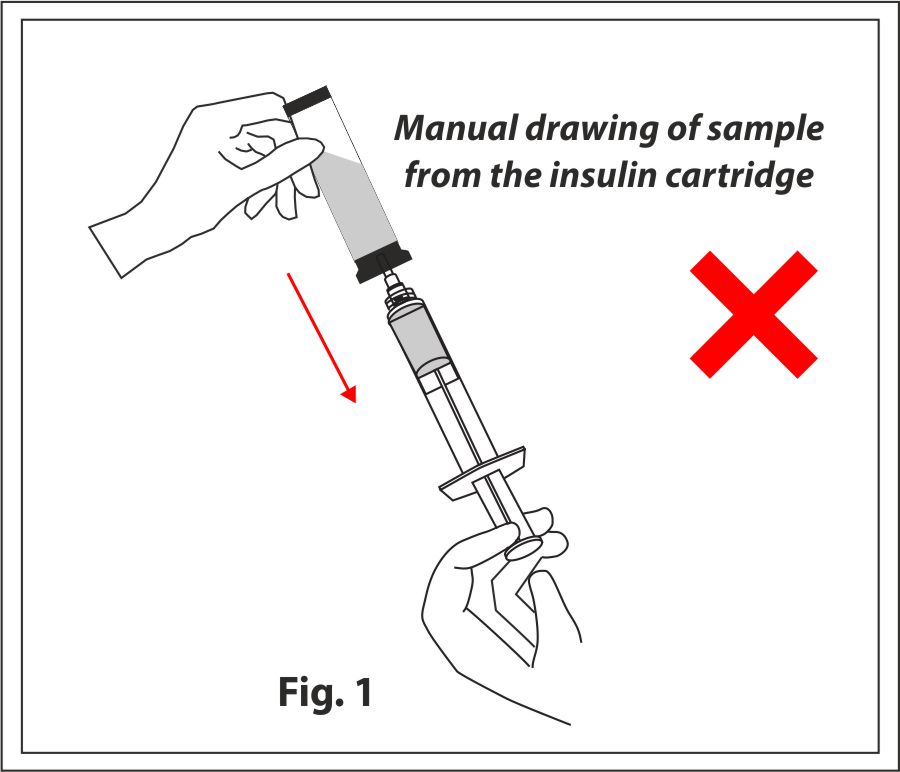
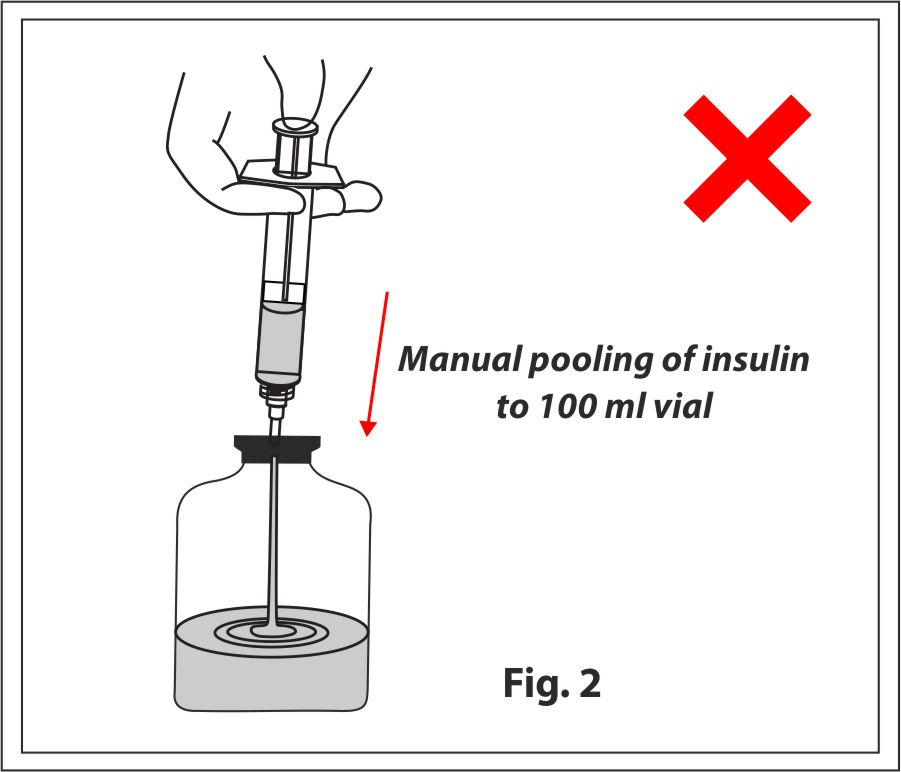
Manual Pooling
A syringe is used to draw the insulin sample from the specialized cartridge and is then pooled into a 100ml vial to be transferred/filtered through the sterility test canisters.
This involves multiple risks and cons such as:
- Chances of extraneous contamination, spillage and manual error
- Increased incidence of false positives
- Increased cost of operation and documentation
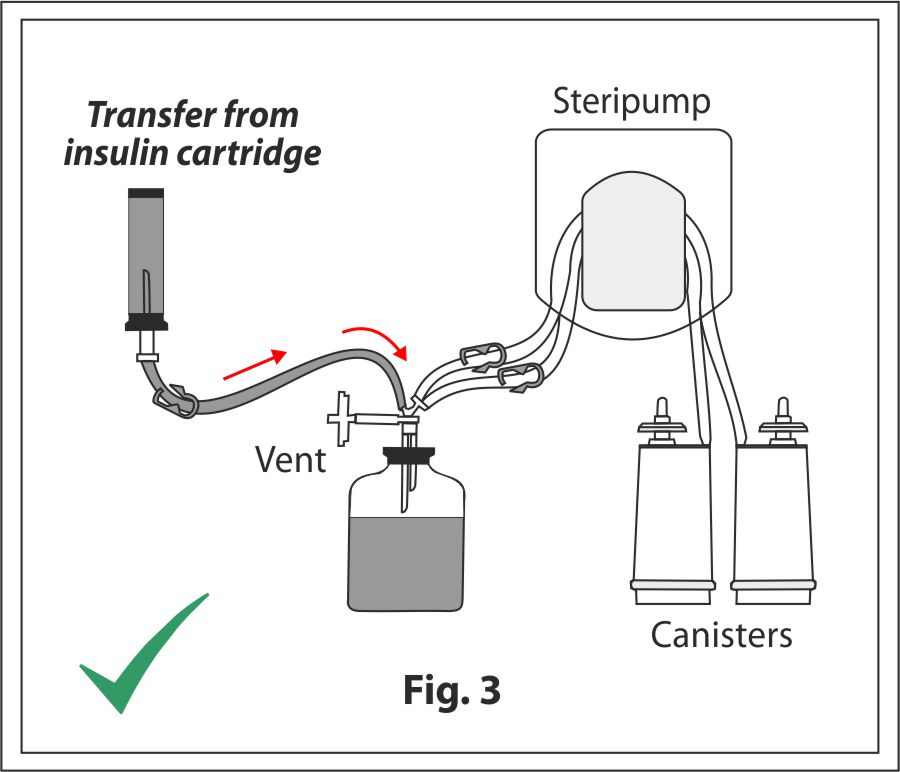
Closed Loop Pooling
A specially designed closed sterility device is now available for closed loop pooling of such special container/cartridges. It obviates the need to use a syringe for pooling the sample thereby minimizing manual intervention and mitigating the risk of extraneous contamination to the test.
Closed loop pooling conducted using Stericheck SVP4 sterility test device for mini vials and cartridges allows swift and efficient transfer of fluid from the cartridge to the pooling vial with easy maneuverability and no additional accessories.